|
3rd Place Essay Competition, 2019 Annual Conference
Using Radiomics to Confirm the Presence of EGFR mutations in Non-small Cell Lung Cancer Patients to Help Increase the Use of Targeted Therapies”
Profige, Kristen
Memorial Sloan Kettering Cancer Center Abstract
Lung cancer is the leading cause of death among men and women around the world. The 5-year survival rate for lung cancer is 15.6%. Finding a subtype, survival rate, stage, and grade of a lung cancer tumor is imperative when choosing a treatment plan. The current standard of care for advanced stage lung cancers has hit a plateau when it comes to better survival rates. The purpose of this paper is to explain how new advances in radiomics and its goal of using targeted therapies as a standard of care will lead to more successful treatment plans in advanced stage non-small cell lung cancer patients. Introduction
Lung cancer is the leading cause of death in the United States, the 5-year survival rate for lung cancer is 15.6%. Patients who are diagnosed at an early stage with localized disease have a better prognosis, approximately a 52% 5-year survival. Although these patients have a better prognosis, it is rare they are diagnosed at such an early stage (1). About 40% of non-small cell lung cancer (NSCLC) patients present with advanced stage lung cancer (2). Choosing a treatment plan for someone with lung cancer is based on survival rates, staging, grade of the tumor, and other specific tumor characteristics. Current Treatment Options for Advanced Stage Non-Small Cell Lung Cancer Patients
According to the American Cancer Society, surgery, chemotherapy, radiation therapy, immunotherapy and targeted therapy can all be used for late stage NSCLC (3). Currently chemotherapy is used prior to surgery or radiation to shrink the tumor or concurrent with radiation therapy to increase radio sensitivity. The most common chemotherapy administered for NSCLC is Cisplatin. The problem with chemotherapy is its inability to distinguish between healthy cells and tumor cells causing it to interact with all cells. This also comes with multiple side effects and can take a toll on patient’s quality of life during and/or after treatment. This is why researchers and doctors are moving towards precision medicine or targeted therapies. These can either be used with or sometimes instead of chemotherapy for better results of treatment. Precision Medicine and Tumor Heterogeneity
Oncology is moving towards individualized medicine or precision medicine, with the discovery of tumor heterogeneity. The rise in genetic sequencing caused us to recognize that each tumor develops differently, reacts differently to treatment, and has different prognostic factors. This is based on the genetic makeup of the individual and certain environmental factors. Intratumor heterogeneity is seen all the way down to the molecular makeup of each individual cell that forms a tumor. Certain biomarkers and phenotypes in an individual’s tumor can tell us a lot about how the tumor will react to targeted therapies and the use of specific drugs. One study states “...medications have demonstrated significant activity within subsets of tumors which harbor specific targetable mutations” (4). With this knowledge we have been finding issues with the way we identify tumor subtypes. We currently use an array of biopsy techniques to view intratumor heterogeneity. One of the issues with this technique is the limited amount of data we receive from one tiny sample of tissue. Intratumor heterogeneity is not only seen in different tumors and that tumor’s tissues but also the individual cells that form the tumor. Recent studies prove that each cell is individualized, and these differences can have a huge impact on the way we approach this tumor. Personalized medicine has helped us lift the yield on targeted therapies Histology and Tumor Heterogeneity of Non-Small Cell Lung Cancer
Lung cancer arises from epithelium cells of the lung and can be either small cell lung cancer (SCLC) or non-small cell lung cancer (NSCLC). NSCLC is the most common histology, about 85% of lung cancers, more specifically non-small cell adenocarcinoma. Due to recent studies on tumor genetics there has been a shift in the way lung cancer is classified (5). Recent studies reveal that 60% of NSCLC cancers express Epidermal Growth Factor Receptor mutations (6). This information has changed the way medical professionals treat some lung cancers due to the sensitivity of EGFR positive tumors to certain drugs. Epidermal Growth Factor Receptor (EGFR)
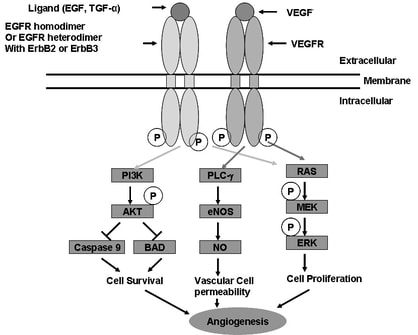
Growth factors in mammalian cells are part of a normal biological process necessary for the development, growth, and proliferation of the cell. They are also a huge asset to osteogenic differentiation in mesenchymal cells. These EGFR are known as “wild type” and are not overexpressed. EGFR belongs to the HER/erbB family of receptor tyrosine kinases and is located on the short arm of chromosome 7. When mutated, the downstream signaling has been shown to lead to increased proliferation, angiogenesis, metastasis, and decreased apoptosis in NSCLC adenocarcinomas (6). More recent studies have shown that lung cancers that have the adeno carcinoma component most likely have the EGFR mutation (7). Figure 1 (8): This figure shows the downstream signaling of EGFR and how it plays a role in cell survival, cell proliferation, and increased angiogenesis.
Radiomics
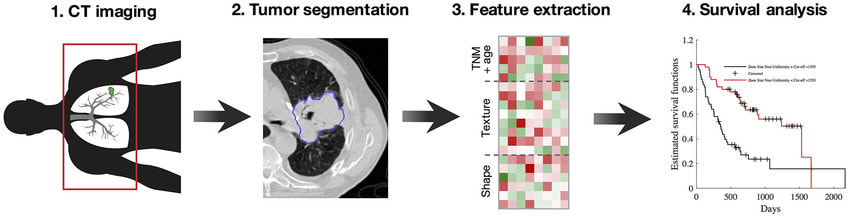
Currently medical imaging is used for the visual aspects of the image and for diagnostic purposes. More recent studies have shown that images are not just what we can see with our eyes. Images are filled with valuable image features and image textures that can be useful in precision medicine. Although there is no concrete definition of radiomics, studies have shown it aims to turn images into mineable and useable data by quantifying characteristic image features. Radiomics is growing quickly based on the fact that cancer patients are imaged as part of their diagnostic workup. This makes it easy to use for not only current patient’s data but all patient’s data from the past. The process of radiomics is complex and can be broken down into four steps. The first step is image acquisition and reconstruction which is taking an image and acquiring the data from that image. The second step is image segmentation and rendering. This involves finding the region of interest which is usually the tumor. The third step is feature extraction and feature quantification. Some features currently being analyzed include: intensity, shape, texture, and wavelet. The final step is to analyze data and create a database (9). This is the biggest challenge in radiomics. There is a huge diversity in image coding and reproducibility due to multiple brands and different types of imaging machines. There is becoming a need for a common lexicon between researchers so that radiomics can become a universal tool. Figure 2 (10): This figure shows a brief workflow of radiomics.
Goals of Radiomics
Currently we get information on tumor heterogeneity from invasive biopsies. However, we can only get biopsies from small samples in random locations. This causes us to fail in accurately representing the full landscape of the biological variation in a tumor (13). Radiomics aims to get the complete landscape of tumor heterogeneity using non-invasive medical imaging that we already took for diagnostic purposes. This will also reduce the cost of cancer staging and grading because we already have medical imaging available and we can skip various invasive tests. Once a universal software is available, doctors from all over the world can collect data on tumor heterogeneity and how each gene in a tumor cell will react to certain treatments and prognostic factors such as recurrence. Not only are we missing the total landscape of the tumor when we only get a sample of tissue, but we also only do a biopsy prior to treatment. Since we know that tumor heterogeneity has an effect on treatment outcome, we need to observe change in tumor heterogeneity before, during, and after treatment for better outcomes. One solution to this is the addition medical imaging along with biopsies. Using Radiomics to Detect the EGFR
Knowing NSCLC treatment outcomes are dependent on tumor heterogeneity motivates us to apply radiomics to the first few steps of treatment planning. According to one study, computed tomography (CT) images showed shape and wavelet features that describe tumor heterogeneity in NSCLC tumors which demonstrated highly proliferating tumor cells (12). These image features were linked to EGFR positive tumors. Another study mentioned that EGFR status can be predicted by 5 radiomic features (11). These include tumor intensity features, textural features, shape feature, wavelet features, and Laplacian and Guassion features. This study had over 700 patients who all had NSCLC adenocarcinomas, some had KRAS-mutated tumors and some had EGFR-mutated tumors. They studied these patients separately and found multiple radiomic features that were associated with EGFR positive tumors. In this study they proved that EGFR positive tumors were more likely to be heterogenous, with a smaller volume, and present and overall lower density on CT images (11). The results of this study show that a large number of NSCLC adenocarcinomas would have successful outcomes with targeted therapies. With the use of radiomics, we can eliminate false negatives in EGFR mutation based on the small biopsy that was taken. |
|
EGFR Inhibiters used for Target Therapy in Non-small Cell Lung Cancer
One study states, despite aggressive surgery and chemotherapy NSCLC is still the leading cause of cancer related death in both men and woman (14). As discussed, this is due to intratumor heterogeneity. Researchers saw this problem and found that targeted therapies were a solution to this problem. Targeted therapies are drugs used to interfere with specific signaling pathways that allow tumor development and growth. EGFR has been seen as an “attractive” therapeutic target in targeted therapies. EGFR is present in highly proliferating NSCLC adenocarcinomas and a negative prognostic factor (14). Researchers have come up with an EGFR inhibitor that is known as EGFR tyrosine kinase inhibitor (TKI). In a recent study done in 2017, they discussed multiple TKIs and their success in the treatment of advanced stage NSCLC patients. The use of TKIs to block the downstream signaling of EGFR positive mutations has shown a better clinical benefit than chemotherapy alone. Gefitinib is one of the first TKIs created as a first-line treatment. This was the first documented trial proving increased progression free survival with the use of TKIs instead of chemotherapy alone. The overall survival was not improved but it did show that targeted EGFR mutations were possible with oral drugs. From there, multiple drugs and trials opened up all finding slight increases in progression free survival. They also tested multiple concurrent treatments such as immunotherapies and antiangiogenic drugs. Although this is an up and coming treatment regimen it shows promising improvements for the overall success in treating advanced NSCLC patients with targeted treatments. They have seen resistance to TKIs and have been improving the targeted therapies as a result (15).
One study states, despite aggressive surgery and chemotherapy NSCLC is still the leading cause of cancer related death in both men and woman (14). As discussed, this is due to intratumor heterogeneity. Researchers saw this problem and found that targeted therapies were a solution to this problem. Targeted therapies are drugs used to interfere with specific signaling pathways that allow tumor development and growth. EGFR has been seen as an “attractive” therapeutic target in targeted therapies. EGFR is present in highly proliferating NSCLC adenocarcinomas and a negative prognostic factor (14). Researchers have come up with an EGFR inhibitor that is known as EGFR tyrosine kinase inhibitor (TKI). In a recent study done in 2017, they discussed multiple TKIs and their success in the treatment of advanced stage NSCLC patients. The use of TKIs to block the downstream signaling of EGFR positive mutations has shown a better clinical benefit than chemotherapy alone. Gefitinib is one of the first TKIs created as a first-line treatment. This was the first documented trial proving increased progression free survival with the use of TKIs instead of chemotherapy alone. The overall survival was not improved but it did show that targeted EGFR mutations were possible with oral drugs. From there, multiple drugs and trials opened up all finding slight increases in progression free survival. They also tested multiple concurrent treatments such as immunotherapies and antiangiogenic drugs. Although this is an up and coming treatment regimen it shows promising improvements for the overall success in treating advanced NSCLC patients with targeted treatments. They have seen resistance to TKIs and have been improving the targeted therapies as a result (15).
Conclusion
Although radiomics and targeted therapies are still being researched, they can have a positive impact on treatment outcomes of NSCLC. Currently biopsies are being used to determine the histology of NSCLC. Radiomics can help us reveal the overall histology and heterogeneity of the tumor. This will increase the chances of patients receiving the best possible treatment outcome. This includes targeted treatments such as TKIs, which have shown success in treating advanced stage NSCLC. With more research and improvement in radiomics and TKIs there will be a huge improvement on survival rates for NSCLC with EGFR positive mutations.
Although radiomics and targeted therapies are still being researched, they can have a positive impact on treatment outcomes of NSCLC. Currently biopsies are being used to determine the histology of NSCLC. Radiomics can help us reveal the overall histology and heterogeneity of the tumor. This will increase the chances of patients receiving the best possible treatment outcome. This includes targeted treatments such as TKIs, which have shown success in treating advanced stage NSCLC. With more research and improvement in radiomics and TKIs there will be a huge improvement on survival rates for NSCLC with EGFR positive mutations.
References
- Dela Cruz CS, Tanoue LT, Matthay RA. Lung cancer: epidemiology, etiology, and prevention. Clinics in chest medicine.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3864624/. Published December 2011.
Accessed March 15, 2019. - Ramalingam S, Belani C. Systemic Chemotherapy for Advanced Non-Small Cell Lung ...
http://theoncologist.alphamedpress.org/content/13/suppl_1/5.full.pdf. Accessed March 18, 2019. - Treatment Choices for Non-Small Cell Lung Cancer, by Stage. American Cancer Society.
https://www.cancer.org/cancer/non-small-cell-lung-cancer/treating/by-stage.html. Accessed March 18, 2019. - Anker CJ, Wo JY. Personalized Medicine in Radiation Oncology—A Work in Progress. International Journal of Radiation Oncology*Biology*Physics. 2015;92(4):843-845. doi:10.1016/j.ijrobp.2015.04.020.
- Cagle PT, Raparia K, Portier BP. Emerging Biomarkers in Personalized Therapy of Lung Cancer. Lung Cancer and Personalized Medicine: Novel Therapies and Clinical Management Advances in Experimental Medicine and Biology. 2015:25-36. doi:10.1007/978-3-319-24932-2_2.
- Santos Gda C, Shepherd FA, Tsao MS. EGFR Mutations and Lung Cancer. Annual Reviews. https://www.annualreviews.org/doi/full/10.1146/annurev-pathol-011110-130206. Accessed March 18, 2019.
- Moreira AL. Diagnosing Non-Small Cell Carcinoma in Small Biopsy and Cytology. Place of publication not identified: Springer-Verlag New York; 2016.
- Zhong H, Bowen JP. Recent Advances in Small Molecule Inhibitors of VEGFR and EGFR Signaling Pathways. http://www.eurekaselect.com. http://www.eurekaselect.com/88155/article. Published May 31, 2011. Accessed March 18, 2019
- Kumar V, Gu Y, Basu S, et al. Radiomics: the process and the challenges. Magnetic Resonance Imaging. 2012;30(9):1234-1248. doi:10.1016/j.mri.2012.06.010.
- Chaddad A, Desrosiers C, Toews M, Abdulkarim B. Predicting survival time of lung cancer patients using radiomic analysis. Oncotarget. 2017;8(61). doi:10.18632/oncotarget.22251.
- Velazquez ER, Parmar C, Liu Y, et al. Somatic Mutations Drive Distinct Imaging Phenotypes in Lung Cancer. Cancer Research. 2017;77(14):3922-3930. doi:10.1158/0008-5472.can-17-0122.
- Chen Y-JY-J, Hua K-L, Hsu C-H, Cheng W-H, Hidayati SC. Computer-aided classification of lung nodules on computed tomography images via deep learning technique. OncoTargets and Therapy. 2015:2015. doi:10.2147/ott.s80733.
- Yip SSF, Aerts HJWL. Applications and limitations of radiomics. Physics in Medicine and Biology. 2016;61(13). doi:10.1088/0031-9155/61/13/r150.
- Sridhar SS, Seymour L, Shepherd FA. Inhibitors of epidermal-growth-factor receptors: a review of clinical research with a focus on non-small-cell lung cancer. The Lancet Oncology. 2003;4(7):397-406. doi:10.1016/s1470-2045(03)01137-9.
- Nan X, Xie C, Yu X, Liu J. EGFR TKI as first-line treatment for patients with advanced EGFR mutation-positive non-small-cell lung cancer. Oncotarget. 2017;8(43). doi:10.18632/oncotarget.20095.