|
2nd Place Essay Competition, 2019 Annual Conference
Magnetic Resonance Imaging: Current Integration and Future Usage in the Treatment of Prostate Cancer in Radiation OncologyShizume-Realmuto, Danica
Memorial Sloan Kettering Cancer Center Abstract
Prostate cancer is one of the most prevalent cancers in men. Current patients diagnosed with prostate cancer have multiple treatment choices. Active surveillance, hormone therapy, chemotherapy, surgery, and radiation therapy are all options available to patients. Patients that elect to receive radiation therapy undergo a simulation prior to treatment. Conventionally, simulation has been done using computed tomography (CT). However, magnetic resonance imaging (MRI) provides high quality imaging in regards to soft tissue contrast. Various studies have shown that synthetic CTs generated from MRI scans are able to be contoured and used for treatment planning. They can also be used to generate digitally reconstructed radiographs (DRR) for therapists to match daily KV images to without significant error. The technology of MRI has not only been introduced and validated in the simulation, but is also showing promise for use in the treatment of numerous cancers in the form of a MRI Linac. Introduction: Prostate Cancer The prostate is a walnut sized gland located in men below the bladder and in front of the rectum. The prostate is responsible for creating and storing seminal fluid that nourishes and transports sperm. Prostate cancer is the most common cancer among men, second only to skin cancer. It is the second leading cause of cancer death in men behind lung cancer. According to the American Cancer Society, it is estimated that in the United States for the year 2019, 174,650 new cases will be diagnosed with 31,620 resulting in death (American Cancer Society, 2019). Prostate cancer symptoms include difficulty urinating, decreased force or weak stream while urinating, hematuria, blood in semen, pelvic discomfort, erectile dysfunction, and bone pain if the cancer has metastasized. Prostate cancer can metastasize to the seminal vesicles, lymph nodes, pelvis, and bones. Patients may show no symptoms of prostate cancer. Instead, the disease may be discovered during the examination of blood work results. Prostate Specific Antigen (PSA) appears in the blood and elevated levels could be an indicator of prostate cancer. Prostate cancer occurs when cells in the prostate become abnormal and develop rapidly. Risk factors for prostate cancer include age, race, family history, and obesity. Prostate cancer is most commonly diagnosed in older men with an average age of 66. African Americans are more at risk for developing prostate cancer while also being diagnosed with more advanced and aggressive cancer. Families with a history of breast cancer and mutations in BRCA1 or BRCA2 also have an increased risk of prostate cancer. Almost all histologies of prostate cancers are adenocarcinoma, with very few being transitional cell, small cell, or a sarcoma. The five year survival rate for localized or regional prostate cancer is almost 100% while distant prostate cancer is 30%. Current Treatment Options Patients diagnosed with prostate cancer have a myriad of treatment options based on the stage of diagnosis. Prostate cancer uses the Tumor, Nodes, and Mets staging system in conjunction with a Gleason score. A Gleason score is determined after the patient has undergone a prostate biopsy. A Gleason score has two grades. The pathologist examines the sample biopsied and determines the most predominant pattern. The most predominant pattern of cells is given a score from 1 (being low) to 5 (being high). The second most predominant pattern is also assessed and given a score using the same scale as the first. Adding these two scores together determines the final Gleason score. The higher the Gleason score, the higher grade the cancer is and the more aggressive it is. Patients with no symptoms, slow growing cancer (based on Gleason score), or small cancer contained to the prostate may not receive treatment. Instead they participate in active surveillance. In active surveillance, patients continue to go to the doctor every six months to a year for bloodwork to check the PSA level. They may also receive digital rectal exams so that the physician can feel if there has been any change in the shape, texture, or size of the prostate. Hormone therapy is another treatment option for patients. The purpose of hormone therapy is to limit the amount of testosterone produced, as testosterone aids cancer cells in dividing. Drugs such as leuprolide, goserelin, triptorelin, or histrelin aim to prevent the testicles from receiving messages to produce testosterone. Other drugs such as bicalutamide, nilutamide, and flutamide prevent testosterone from reaching the cancer cells. Patients could have a surgical procedure called an orchiectomy to remove the testicles and therefore reduce testosterone as well. Other options include surgery, such as a radical prostatectomy, chemotherapy for systemic disease, or radiation therapy. Radiation therapy can be delivered via external beam or brachytherapy. An external beam radiation therapy treatment can be delivered in many ways. Patient treatment plans can be created as a 3D conformal plan, an IMRT/IGRT plan, a SBRT plan, a proton therapy plan, or a VMAT plan. Whatever the plan chosen, all patients who elect to have radiation therapy treatments undergo a simulation. Computed Tomography Simulation and Treatment Planning The most common simulation to date for prostate cancer has been a CT simulation after fiducial placement. During this simulation process, the patient is introduced to and set up in the position that they will be receiving treatment. An immobilization device, typically a thermoplastic mold, is made over the pelvis and scans are taken. These scans are used by the treatment planning team to develop an accurate and individualized plan for the patient. Patients also receive permanent markings, or tattoos, to assist the radiation therapist in accurate set ups for treatment. CT scanners are calibrated in relation to the radiodensity of water and air at standard temperature and pressure. Hounsfield Units are used to help determine the density of different tissues, bones, and liquid in the body (Greenway, 2019). The Hounsfield unit is a linear transformation of the measured attenuation coefficients. The water in which the machine is calibrated to has a Hounsfield unit of zero and air -1000. Hounsfield units are allocated differently to tissues and bones. For example, bone has a Hounsfield unit range up to +2000 while an organ such as the lungs is -700 to -600. These units are important in treatment planning because they have the ability to measure the density of a region of interest and for accurate calculations of tissue inhomogeneities. The introduction of MRI in radiation oncology simulation poses a challenge with the absence of electron density information. Magnetic Resonance Imaging: How it Works
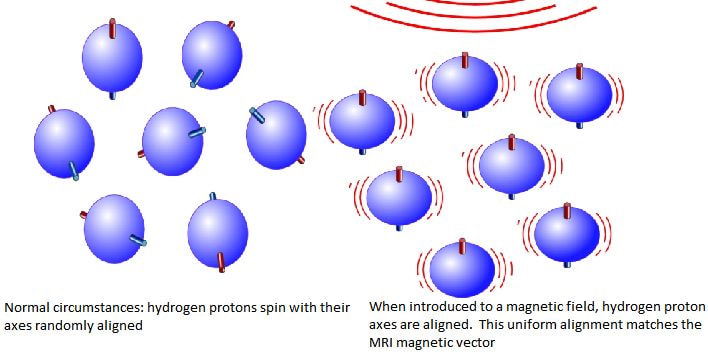
An MRI scanner works by using the body’s natural magnetic properties to produce detailed images (Berger, 2019). Disease often manifests by increasing water. MRI is sensitive to this detection. MRI targets the hydrogen nuclei protons in the body. These hydrogen nuclei are abundant in both water and fat. The protons of these molecules spin on an axis with a north-south pole orientation. Under normal conditions within the body, these protons spin on random axes. During an MRI scan, these protons are aligned in the magnetic field, matching the axis of the scanner. A radiofrequency wave is produced by the scanner which stimulates the protons and causes them to spin out of equilibrium. When this radiofrequency wave is turned off, the protons return to the alignment of the magnet. During this process, another radiofrequency wave is emitted and this signal is used to produce images. Receiver coils are used to act as aerials to improve the detection of the emitted signal. Different tissues return to their normal state at different rates and have different relaxation times. This helps the physician distinguish different tissue types based on the magnetic properties. Proton relaxation can be seen in two ways. The first way is T1 relaxation, which is the time taken for the magnetic vector to return to the resting state. The second way is T2 relaxation. T2 relaxation is the time needed for the axial spin of the protons to return to the resting state. Diffusion weighted images are taken to see how water molecules diffuse through the body. Fat suppression sequences remove the signal from fat, leaving only the signal from any abnormalities in the area. The strength of the magnetic field could be altered using different electric coils, different frequencies, or different slice numbers. Contrast agents, like Gadolinium, increase the speed at which the protons realign. The faster the protons realign, the brighter the image. Although CT scans do provide better bony anatomy images, the soft tissue contrast is exceptional on an MRI. However, there are disadvantages and limitations to MRI. MRI scans have a longer duration than CT scans, though in MRI there is no exposure to ionizing radiation. MRI also tends to be more expensive than CT. Limitations to MRI are patients with pacemakers or certain metal or cochlear implants. A patient with these limitations may not be eligible for MRI and therefore undergo a CT simulation.
MRI Simulation and Synthetic CT
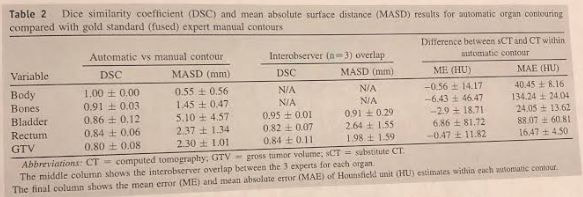
MRI provides superior soft tissue contrast compared to CT while also providing anatomical and functional information of the scanned area. While an MRI and CT fusion is possible to attempt to overcome the missing electron density information, current studies are exploring the feasibility of an MRI only simulation for prostate cancer. MRI simulation consists of an interdisciplinary team in which MRI technicians, MRI physicists, and radiation therapist work together (Rai et al., 2017). An MRI scan provides better tumor visualization and better visualization of nearby critical structures. This is important for treatment planning so that organs at risk can be avoided while still accurately delivering the desired dose to the target. While diagnostic MRI scanners have a curved table, it is extremely important for the table to be flat in an MRI simulation since tables are flat in CT (if a fusion) and in the treatment room. The flat table also improves reproducibility and increased accuracy of image registration for the treatment planning team. MRI simulations must be performed with any immobilization device that is going to be used in treatment. Large immobilization devices could be challenging as they need to be able to fit inside the bore of the machine. It is extremely important that the patient be as still as possible, as MRI images are sensitive to motion and image quality for volume delineation is critical. A recent study explored the accuracy of synthetic CT scans and atlas based registration in regards to prostate cancer simulation (Farjam et al., 2018). Concerns were noted regarding the misregistration between MR and CT scans and the possible differences in bladder and rectal filling. However, with MR being the primary method for radiation therapy planning and synthesizing a CT from the MRI information, this concern is eliminated. This study explored MR for calculating attenuation, or MRCAT, and synthetic CT image registration using deformable registration. MRCAT is performed using a 3T magnet with three distinct images: water only, fat only, and in phase MRI. The classification algorithm used provides soft tissue and bony clusters which are then divided into water, adipose tissue, cortical, and spongy bone. They are then assigned a bulk electron density. This bulk electron density is to account for the missing Hounsfield units found on CT only. This protocol is currently limited to the manufacturer and scans are limited to generate bone only up to the L4 level. This limitation is not ideal for patients who have nodal involvement higher than L4. In this study, 23 sets of CT and MR images were retrospectively selected from a pool of prostate cancer patients ranging in age from 54-87 years old. These patients also had MRCAT scans along with CT scans and MR separate scans. All scans were performed in the treatment position. The MR scans had both anterior and posterior coils and 2.5mm slices. CT slices were also 2.5mm. The purpose of synthetic CT generation is to assign a CT number, or Hounsfield unit, to each MR voxel. Because the MR image is of better quality, images from CT were deformed onto the MRI. Water only images were used primarily to obtain superior image contrast between bone and soft tissue. These images also aided in the deformable image registration between CT and MR. The study showed the largest discrepancy between synthetic CT and deformed planning was in the bony structures. In this study, synthetic CT was slightly superior in terms of Hounsfield unit assignment. The MRCAT performed more efficiently with very sharp and clean images, whereas the atlas based deformable images were slightly blurred. This can pose challenges during treatment planning and contouring of critical structures. This study concluded that the multi atlas approaches outperforms MRCAT with Hounsfield unit allocation. It also does slightly better in terms of dose distribution. The slight blurriness of the image though still poses a challenge for treatment planning. A significant barrier to MRI only workflow is that scans cannot be calibrated to electron density due to different imaging principles between CT and MRI. Therefore dose calculations cannot be performed. A separate study also explored synthetic CT scans in relation to prostate cancer (Dowling et al., 2015). This study enrolled 39 patients with localized prostate cancer. Patients were between the ages of 58-78 years old and were diagnosed between stages T1-T3. All patients had three gold fiducials placed one to two weeks prior to simulation. Patients received a whole pelvis MRI T2 weighted scan and a routine planning CT scan. Contouring of the prostate, rectum, bladder, and bones was done manually on the MRI and the CT scan. Each CT scan was coregistered to the T2 whole pelvis MRI. Registration and atlas based approaches need deformable mapping of the CT intensities onto the MRI. Registration of each atlas MR to the target MR followed by a fusion of the coregistered CT to estimate the Hounsfield units improves accuracy. Patients were simulated in CT with a full bladder and empty rectum. The MRI scanner had a dedicated flat couch and coil mounts. Patients were positioned by two radiation therapists for the MRI. Patients were to have a full bladder and empty rectum for the MRI as well. The field of view for the MRI was to cover the entire pelvis, including all of the bladder. All images were uploaded to a treatment planning software.
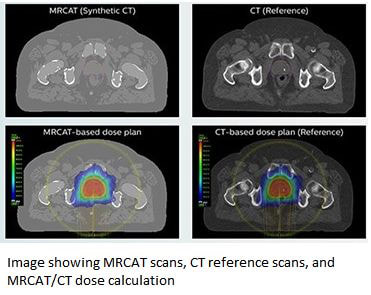
The above graph shows that substitute CT scans can be automatically generated for MRI using T2 weighted sequences and dose calculations and contours are comparable to conventional CT. The MRI simulation set up and automatic synthetic CT generation creates realistic contours and electron densities for prostate cancer radiation dose planning.
Another study performed assessed the feasibility of an MRI only workflow for prostate cancer (Tyagi et al., 2017). Again, MRI has superior soft tissue contrast which improves delineation of the prostate and adjacent normal tissues compared to CT. According to this study, using CT alone overestimated the prostatic volume by 30-40%. A flat table with indexing was utilized as well as a laser positioning system. MRI as the primary simulation aims to minimize dosimetric error that is introduced by the misregistration of CT and MR images. It also seeks to minimize the possible changes in anatomy, such as bladder or rectal filling between having the two scans performed, and to reduce redundant imaging and patient inconveniences. To achieve an MRI only workflow, a synthetic CT generated form MR image sets is needed. MRI isocenter marking is also needed, as is sufficient contrast for treatment planning contouring. 2D digital reconstructed radiographs or 3D reference images are also required. This study had 48 patients who were being treated to an intact prostate gland or the postoperative prostate bed. Patients were to receive external beam radiation therapy or a boost to the area following brachytherapy. The MRI set up in this study matched the institutions current set up for CT simulation. A thermoplastic immobilization device was to be made on a pelvic board and the patients would be receiving permanent markings. The MRI table was a flat table which housed the posterior coils. The posterior coil placement was located 1cm closer to the patient which is similar to a diagnostic table. The flat table had a pelvic board on top so that the immobilization device could be worn during MRI. For patients with an intact prostate gland, three fiducials were placed in the prostate prior to simulation. These fiducials are used to monitor the prostate position. Patients who were to receive stereotactic body radiation therapy also received a rectal spacer. Patients in this study first went to CT simulation for the mold to be made and tattoos placed. Measurements were also done at this time to ensure that patients would be eligible for MRCAT sequences. If patients were not eligible (exceeding 50 cm left to right and 30 cm anterior to posterior), patients had a full CT simulation. Patients with hip prosthesis were also excluded from continuing as the prosthesis affects accurate classification of bone tissue on MRCAT. Patients that were eligible for MRCAT after the immobilization device was made had a low resolution MRI scan to assess bladder and rectal filling. Any gas in the rectum was removed with a catheter prior to continuing. Patients were then positioned with the immobilization device to the permanent markings. MRI compatible radio opaque markers were placed on the patients tattoos. The markers are placed so that isocenter planning is accurate in regards to triangulation points. An anterior coil is placed on top of the immobilization device. Of the 48 patients enrolled in this trial, 42 patients completed the MRCAT MRI only simulation. 4 patients had hip prosthesis and 2 patients exceeded the measurement limits. 42 patients were successfully simulated, planned, and treated with MR only workflow. Two of these cases were SBRT while eleven were salvage therapy receiving 1.8Gy for 40 fractions. An average of 15 minutes was saved using MRI only simulation versus a combined CT and MRI simulation. Physicians were able to successfully contour the CTV (prostate and seminal vesicles), bladder, urethra, and rectum. Contouring on MRI only was quicker, easier, and more accurate when compared to CT+MRI simulations. MRCAT patients had a 2D bony DRR match for prostate bed patients or a 2D fiducial match with a 3D CBCT intrafraction motion review for intact prostates. This study demonstrated that an MRI only workflow is feasible and can achieve similar geometric and dosimetric accuracy as CT alone or CT+MRI simulations. A different study analyzed if image guided radiation therapy (IGRT) was achievable using MRI only images from simulation (Kemppainen et al., 2018). A comparison was made between CT and MRI digitally reconstructed radiographs using daily KV imaging. In contemporary external beam radiation therapy of the prostate, CT is the primary imaging source with MRI being supplementary. The drawback of having a multi modality imaging system for simulation is the possibility of misregistration of images. Using MRI only eliminates this problem. This study had 20 participants who were treated with external beam radiation therapy based on a CT simulation. Additional MRI images were registered with the planning CT for target delineation and organs at risk contouring. Secondary MRI images, using the MRCAT sequence, were also taken allowing a retrospective comparison between CT and MRI digitally reconstructed radiographs. DRRs are important for IGRT because they verify a patient’s position prior to treatment and ensure accurate dose delivery. For the secondary MRI, patients were positioned with the same immobilization and position from the CT simulation. A flat table was once again used and an anterior coil was placed on the patient. Two orthogonal KV image pairs were taken using the linear accelerators on board imaging. Five observers (4 radiographers and 1 physicist) manually registered the CT and MRCAT based DRRs to the daily KV images. Each pair was evaluated three times by the same observer to obtain repeated measurements. This enabled the assessment of measurement quality for inter and intra-observer variability. The longitude, latitude, and vertical table coordinates were evaluated in both the CT and MRCAT KV match. The observers matching were not aware if they were matching to the CT DRR or the MRCAT DRR. The inter-observer variation was not significant in the longitude or the latitude, but the vertical was statistically significant. The lateral was almost identical in both, with slight deviation in the longitude. However, the intra-observer variation was significant in all three directions, the largest being longitudinal and the smallest latitudinal. The average difference in the vertical was -0.5, the longitudinal 0.1, and the latitudinal -0.1. The study concluded that the accuracy of the MRCAT DRRs were clinically acceptable. Future studies performed need to investigate the use of MRCAT with cone beam CT (CBCT) based position verification utilizing fiducial placement. This is of clinical interest as MRCAT only has two soft tissue density values and might not be able to be registered to a CBCT. Since CBCTs have become a common part of IGRT prostate treatments, clinical studies are in need to explore the possibility of furthering MRI only simulations. MRI Linac and Future Potential
Radiation therapy has evolved exponentially throughout the years, from MV images to now CBCT and intrafraction motion review (IMR). However, CBCT scans have poor soft tissue contrast and have difficulty capturing organ motion, even with respiratory gating. Integrating a MRI with a linear accelerator eliminates this while not exposing a patient to excess ionizing radiation. MRI imaging can capture both anatomical and functional information and is the next step in improving real time images during treatment. Diffusion weighted image sequences are particularly valuable. These sequences evaluate the movement of water protons and track molecular activity. This movement may be restricted in dense cell structures of tumors. Therefore, real time DWI imaging can help assess a tumor during treatment (Zagoudis, 2019). Merging MRI and linear accelerators is still early in development and practice. The goal of this machine is to use the MRI portion to relay high quality, real time images as a patient is being treated with radiation from the linac portion of the machine. In a current model being used at MD Anderson, the MRI Linac splits the metal coil in half so that there is a gap in the middle of the machine where the patient is positioned (Wendler, 2019). This allows radiation to pass through unmagnified and images are created without distortion. If the tumor moves out of the target zone due to motion or breathing, the radiation automatically shuts off. It automatically resumes treatment once the target has returned to the target zone. This process spares healthy tissue from receiving dose and allows physicians to plan and deliver a more powerful dose to the target. Because MRI Linac is still in its early state, not much research has been done specific to one treatment site. One study did explore the ability of using a 1.5T MRI Linac for prostate cancer (Kooreman et al., 2019). In this study, 4 quantitative MRI sequences were performed: T1 mapping, T2 mapping, DWI, and dynamic contrast enhanced. These sequences were first performed on phantoms and looked at the short and long term repeatability and reproducibility. All quantitative MRI sequences were then performed on a single prostate cancer patient. There was not much variability within the sequences in regard to short or long term repeatability or reproducibility. The study demonstrated the feasibility of quantitative acquisition in a patient with prostate cancer. The accuracy and repeatability for T1 and T2 were similar to the literature values for a diagnostic system. This suggests that the MRI Linac performs similar to the diagnostic MRI and can be used for treatment response monitoring as well with high quality images. While not prostate cancer, a different study was performed with a 1.5T MRI Linac for patients with bone metastases (Raaymakers et al., 2017). Four patients with lumbar spine bone metastasis were treated with a three or five been step and shoot IMRT plan. The plan was created while the patient was on the treatment table based on the real time MRI images. A CT taken before treatment was registered to this MRI to assign Hounsfield units. A single fraction of 8Gy was prescribed. The MRI Linac used in this study could only move the table in a longitudinal direction. Patients were set up to lasers in the room. However, patient setup was not as important as it is in contemporary treatment. This is because the real time MRI displays the patient's tumor and anatomy and any adjustments could be made based off real time scans. After being assessed by the physician, new plans are able to be created, again based off current real time scans. The table contained a posterior coil. The anterior coil is placed floating above the patient and is radiation transparent, though no beams entered through it. The workflow of the machine was automated with only critical decisions delegated to the operator. A contouring program was continually running as well. A plan is generated from the MRI taken after the patient is setup. The radiation oncologist and physicists review the plan before proceeding. Once the plan was approved, another MRI was taken to ensure there was no movement and the contours are still in place. When treatment began to be delivered, the MV panel is activated to capture portal images for offline validation. A seven second MRI was acquired and presented every seven seconds for continuous patient monitoring. All four patients treated in this manner received palliative effect. The average time for treatment was 41 minutes and was tolerated well by all four patients. The time to beam on, imaging, contouring, and online planning took an average of 25 minutes. Promising studies like these mentioned demonstrate that MRI Linac treatments that meet clinical and departmental goals are achievable. |
|
Conclusion
Excluding skin, prostate cancer is the most common cancer among men. Although the five year survival rate for localized prostate cancer is almost 100% with current treatment options, scientists are trying to integrate new options into the department of radiation oncology. With the introduction of magnetic resonance imaging to radiotherapy, physicians will be able to delineate gross tumor volume, planning target volumes, and organs at risk definitively and precise. This is important in the treatment of prostate cancer since critical structures such as the bladder, urethra, and rectum all lie within the area. A collection of studies discussed in this paper validate the use of MRI both in simulation and in treatment. A multidisciplinary team of radiation oncologists, physicists, radiation therapists, and MRI technicians is needed to ensure that the safest and most accurate conditions are met. Although MRI Linac is still in its early stages of implementation, the technology shows promise in treating various cancers with real time imaging, adjustable treatment planning, and highly accurate dose distribution. The superior soft tissue contrast assists physicians and treatment planners in avoiding organs at risk while delivering powerful doses to the target. Less side effects may be experienced and patients are exposed to less ionizing radiation, forgoing the traditional CT simulation and current treatment monitoring options. MRI is not only the next step in the simulation of patients, but also in the ever advancing image guided radiation therapy treatment process.
Excluding skin, prostate cancer is the most common cancer among men. Although the five year survival rate for localized prostate cancer is almost 100% with current treatment options, scientists are trying to integrate new options into the department of radiation oncology. With the introduction of magnetic resonance imaging to radiotherapy, physicians will be able to delineate gross tumor volume, planning target volumes, and organs at risk definitively and precise. This is important in the treatment of prostate cancer since critical structures such as the bladder, urethra, and rectum all lie within the area. A collection of studies discussed in this paper validate the use of MRI both in simulation and in treatment. A multidisciplinary team of radiation oncologists, physicists, radiation therapists, and MRI technicians is needed to ensure that the safest and most accurate conditions are met. Although MRI Linac is still in its early stages of implementation, the technology shows promise in treating various cancers with real time imaging, adjustable treatment planning, and highly accurate dose distribution. The superior soft tissue contrast assists physicians and treatment planners in avoiding organs at risk while delivering powerful doses to the target. Less side effects may be experienced and patients are exposed to less ionizing radiation, forgoing the traditional CT simulation and current treatment monitoring options. MRI is not only the next step in the simulation of patients, but also in the ever advancing image guided radiation therapy treatment process.